|
We are also
interested in using carbohydrates as ligands for metal-catalyzed
enantioselective syntheses. For this purpose we develop syntheses of novel
sugar derived ligands which are suitable for transition metal mediated CC
bond formations.
Enantioselective Tsuji-Trost Reaction
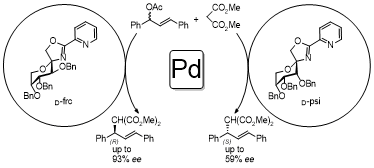
Chiral Iodonium Catalysts
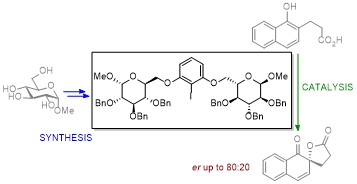
We also synthezise novel sugar structures related to naturally
occurring compounds like peltalosa or rhizobial signal substances
Decadiuloses of the Peltalosa Type
  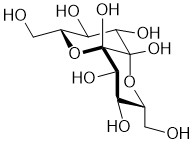 
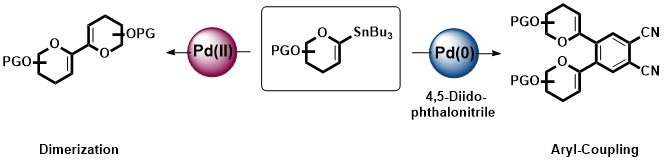
Tributylstannyl Saccharides
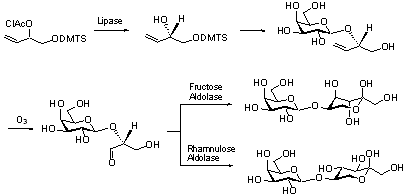
Enzymatic Syntheses
We use enzymes like lipases and aldolases for the synthesis
of enatiopur compounds and sacchardies which are otherwise difficult to
prepare. For example, disaccharides and conduritols were prepared
chemoenzymatically this way.
 |
![]()